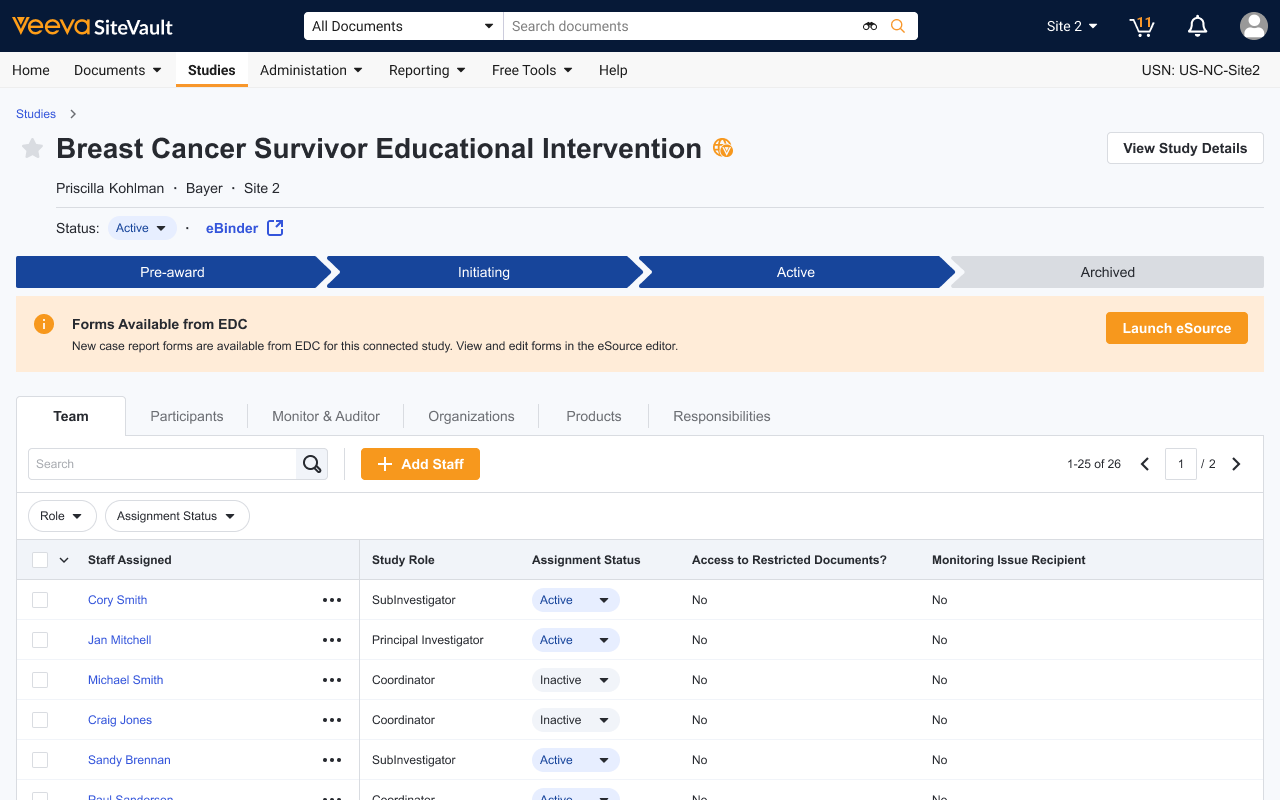
Benefits for Sites
How eSource will change site operations
Eliminate manual data re-entry
Between source data and sponsor EDC
Accelerate Reimbursement
With faster, more accurate EDC submissions
Increase Data Quality
With automated form creation, data checks, and straight through data streaming
Maximize Efficiency
Less time entering data and more time with patients
Benefits for Sponsors
How eSource will enhance collaboration
Faster, Higher Quality Data
Eliminate Source Verification
Reduce Queries to Sites
Frequently Asked Questions (FAQs)
How will Veeva eSource differ from other eSource technology?
Veeva eSource is designed for all site types as a product within Veeva SiteVault, built to work alongside SiteVault eISF, eConsent, and CTMS - and with native interoperability with EHRs and EDC. No bolt-on products required.
Does a sponsor need to have Veeva EDC for my site to benefit from Veeva eSource?
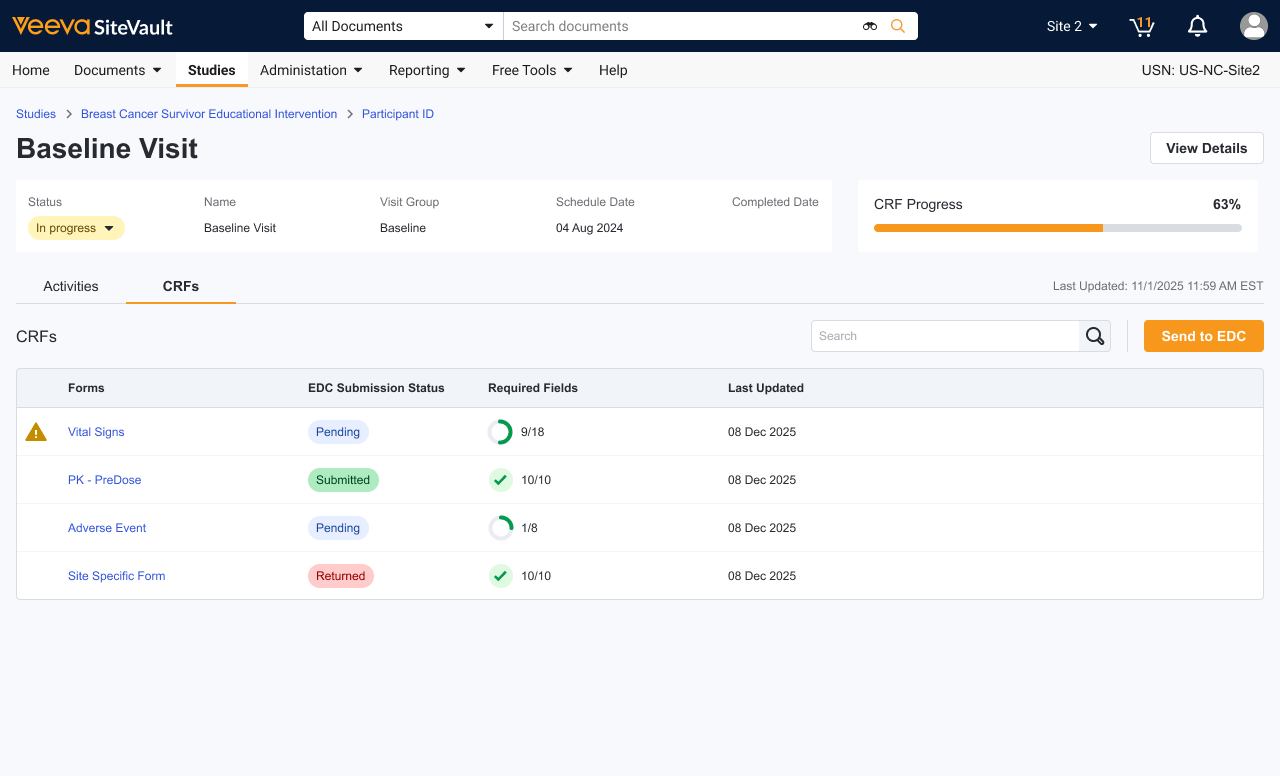
Veeva eSource can be used no matter the EDC, with emphasis on integration built using a standard industry format. There are added benefits when connected to Veeva EDC, like robust bidirectional data flow and increased automation.
What EHR systems can be connected?
Veeva eSource can connect with any major EHR that conforms to global industry standards. It has been tested with large EHRs including Epic. For non-FHIR systems, or systems with unique needs, Veeva is happy to partner with site EHR teams to support.
Can Veeva eSource be used even without an EHR (i.e. if a site uses paper charts, not EHR)
Yes. The value in Veeva eSource is going digital, saving time, trending data, and accessing source anywhere, in addition to the connectivity to the sponsor's EDC.
How will Veeva eSource address sharing identifiable information (PHI,PII) between sites and sponsors?
Veeva eSource is compliant with all global standards for identifiable patient data, and only communicates the appropriate de-identified data information with sponsors & CROs.
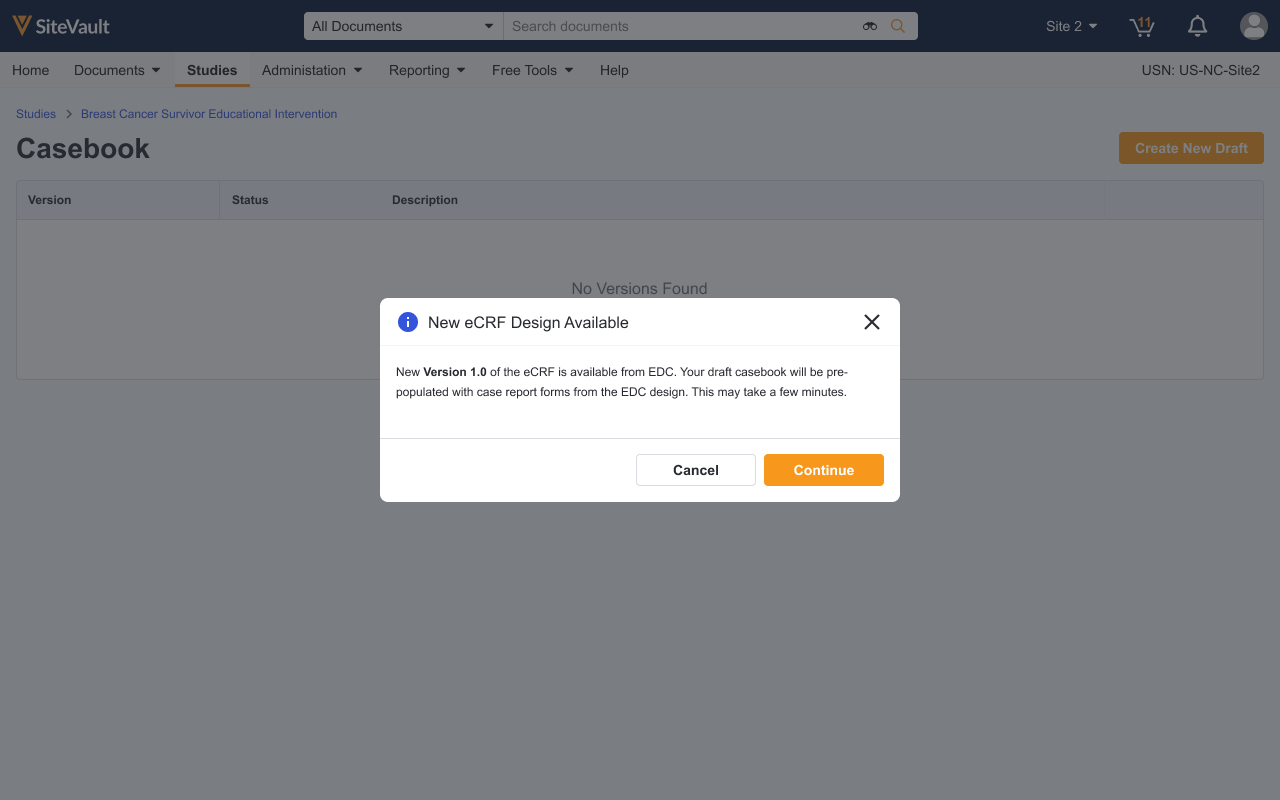
If the EDC database is updated (i.e. a protocol amendment), how does this flow over to Veeva eSource?
As casebook versions are updated in EDC, notifications will be provided in Veeva eSource that changes have been made. Site users can see, download, and easily make these changes for upcoming patient visits. This helps keep sites more aligned with protocol changes happening in EDC.
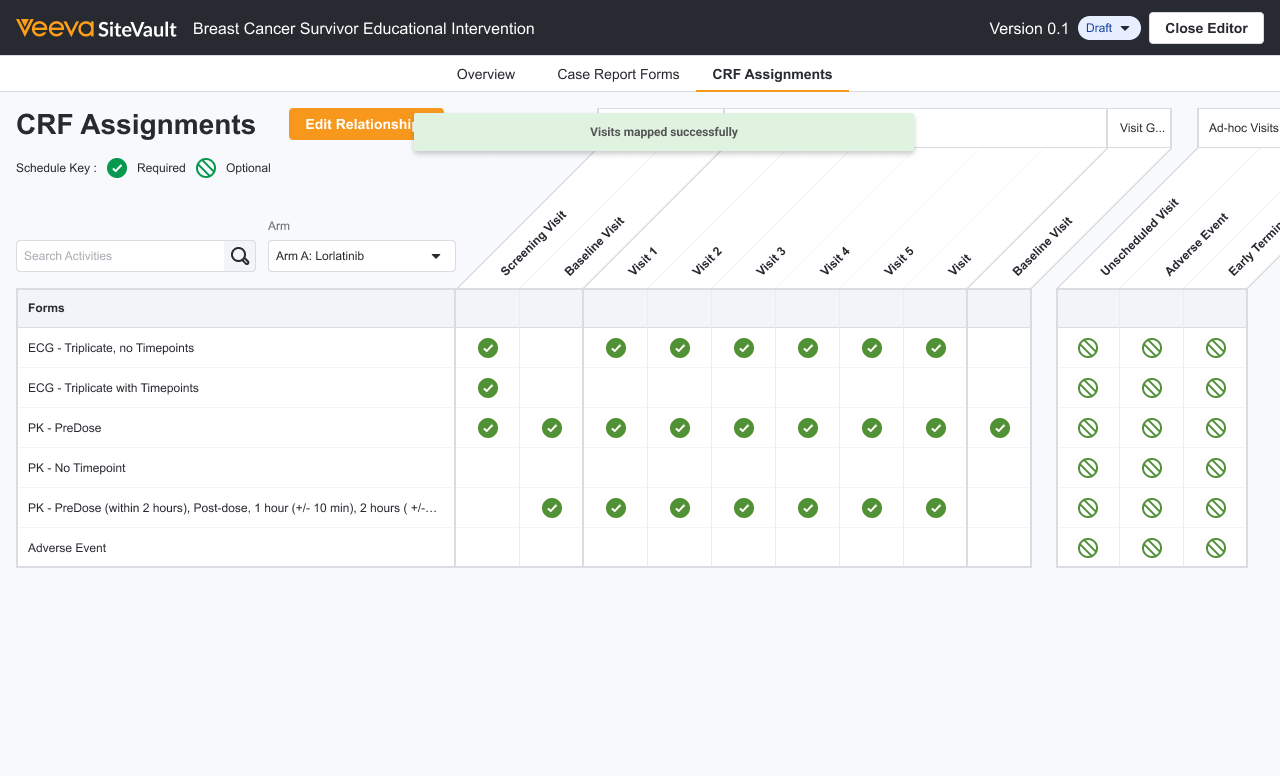
Can sites add their own data or use site-specific forms?
Veeva eSource offers the flexibility to utilize both sponsor-provided eCRFs directly from EDC and custom, site-specific forms. This hybrid approach ensures seamless data flow while accommodating the unique clinical workflows and diverse requirements of every study site.
What are the costs for sponsors and sites?
Veeva SiteVault, which includes Veeva eSource, is free to use for sites with 20 or fewer active studies annually. Sites using Veeva SiteVault Enterprise will be able to license Veeva eSource. As a site-owned product, Veeva eSource is licensed by the site.
eSource will be available to sites using SiteVault CTMS
That means it will be FREE to use for SiteVault CTMS organizations with 20 studies or less,
and available to license for enterprise CTMS organizations with more than 20 active studies.
Integrations, including EMR integrations, are only available with an enterprise license.
Get Started with CTMS or eISF today

.png)
